 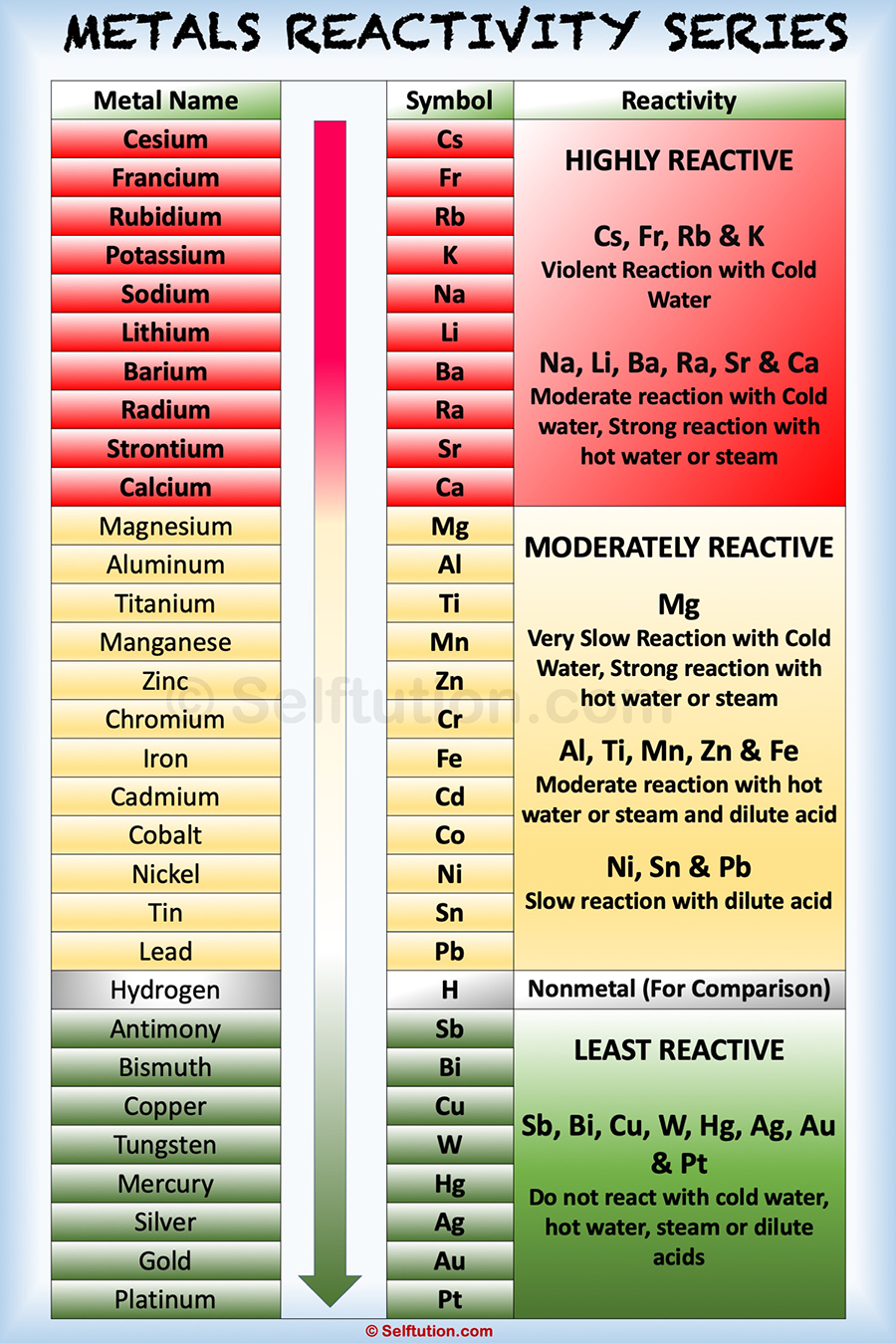
The mnemonic to remember the reactivity series of metal is : Please send cats, monkeys and zebras in lovely happy cages made of silver, gold & platinum. A more sophisticated calculation involving electrode potentials is required to make accurate predictions in this area.\): Elements categorized into metals, non-metals and metalloids. Lesson Two The Reactivity Series 2 Introduction The Periodic Table (found at Appendix A at the back of this folder) lists the 100+ chemical elements. The reactivity series is the series of metals arranged in decreasing order of their reactivity. These rows contain elements in the lanthanoid and actinoid series, usually from 57 to 71 ( lanthanum to lutetium ) and 89 to 103 ( actinium to lawrencium ), respectively. 
The metals which show the fastest and most violent reactions are the most. The periodic table has two rows at the bottom that are usually split out from the main body of the table. For example, calcium is quite reactive with water, whereas magnesium does not react with cold water but does displace hydrogen from steam. A reactivity series of metals can be created using the observations of their reactions with oxygen, water and acid. The boundary between the metals that react with water and those that don't is harder to spot. The table shows four metals into an order of reactivity (from most reactive to least reactive) based on the observations of how the metals react with dilute hydrochloric acid. Those metals that can displace H + ions from acids are easily recognized by their position above H in the activity series. 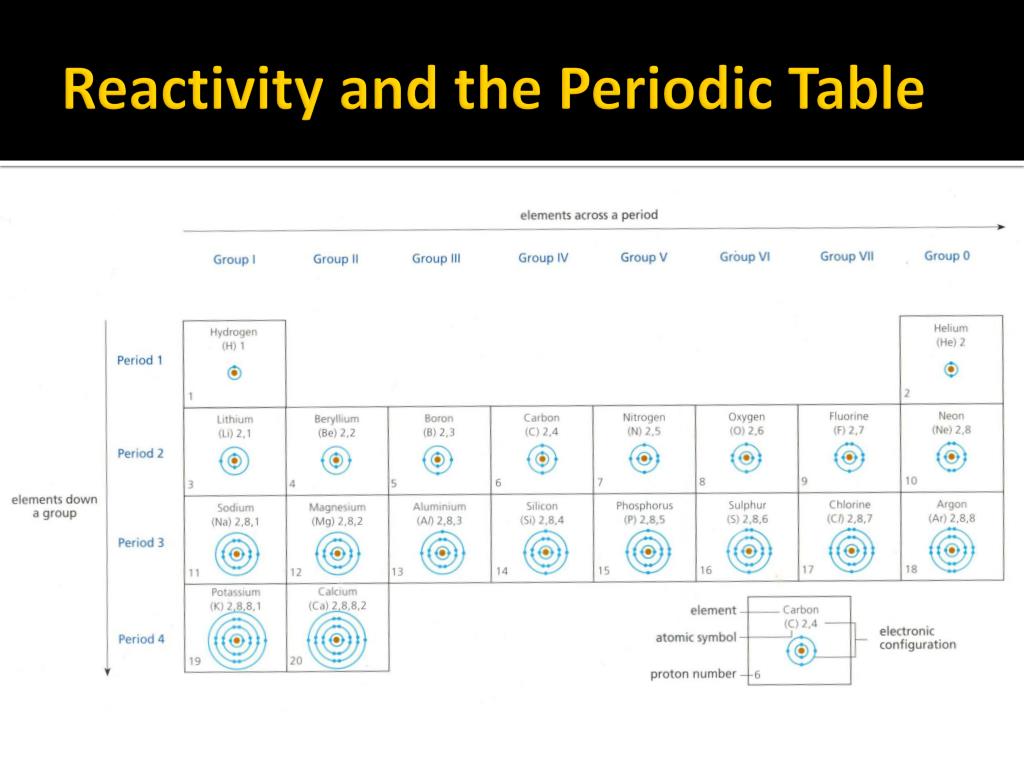
Activity series of some of the more common metals, listed in. It is used to determine the products of single displacement reactions, whereby metal A will replace another metal B in a solution if A is higher in the series. The activity series is a list of elements in decreasing order of their reactivity. Therefore, it is useful to have a list of elements in order of their relative reactivity. Less active metals like iron or zinc cannot displace hydrogen from water but do readily react with acids: The reactivity series is a series of metals, in order of reactivity from highest to lowest. Single-replacement reactions only occur when the element that is doing the replacing is more reactive than the element that is being replaced. Sodium is highly active and is able to displace hydrogen from water: The Group 1 elements, also known as the alkali metals, all react vigorously with water to produce an alkaline solution. Metals are found on the left and in the middle, whereas. 
For example, sodium due to its high reactivity explodes as came in contact with air or water, thus needs to be kept away from moisture and air, and dipped. These metals can get tarnished or corrode very easily. 1, reacts readily with metals to form compounds that can be broadly classified as salts and, therefore, are known as the halogens, which is derived from a combination of Greek words that translate to 'salt makers. 2.1.1 recall the reactivity series of metals, including K, Na, Ca, Mg, Al, Zn, Fe and Cu 2.1. GCSE WJEC Group 1 alkali metals Explaining reactivity. The periodic table can be used to find out if an element is a metal or a non-metal. The reducing tendency of metals at the top of the table has high, which is why they are easily oxidized. It is important to distinguish between the displacement of hydrogen from an acid and hydrogen from water. Each of the elements found in this column, which are boxed in lavender in Figure 2.2.1 2.2. The activity series of metals or reactivity series is a list of metals from most reactive to least reactive. However, silver cannot displace copper ions from solution. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
